Nov 15, 2023, 5:00 am UTC
4 min
Created by
The "protein-misfolding problem": How some neurodegenerative disorders intersect
This article covers:
- Why do proteins misfold?
- How does misfolding cause neurodegeneration?
- Protein misfolding in:
- Huntington's disease
- Alzheimer's disease
- Parkinson's disease
- Amyotrophic lateral sclerosis (ALS)
- Can protein misfolding and its consequences be stopped?
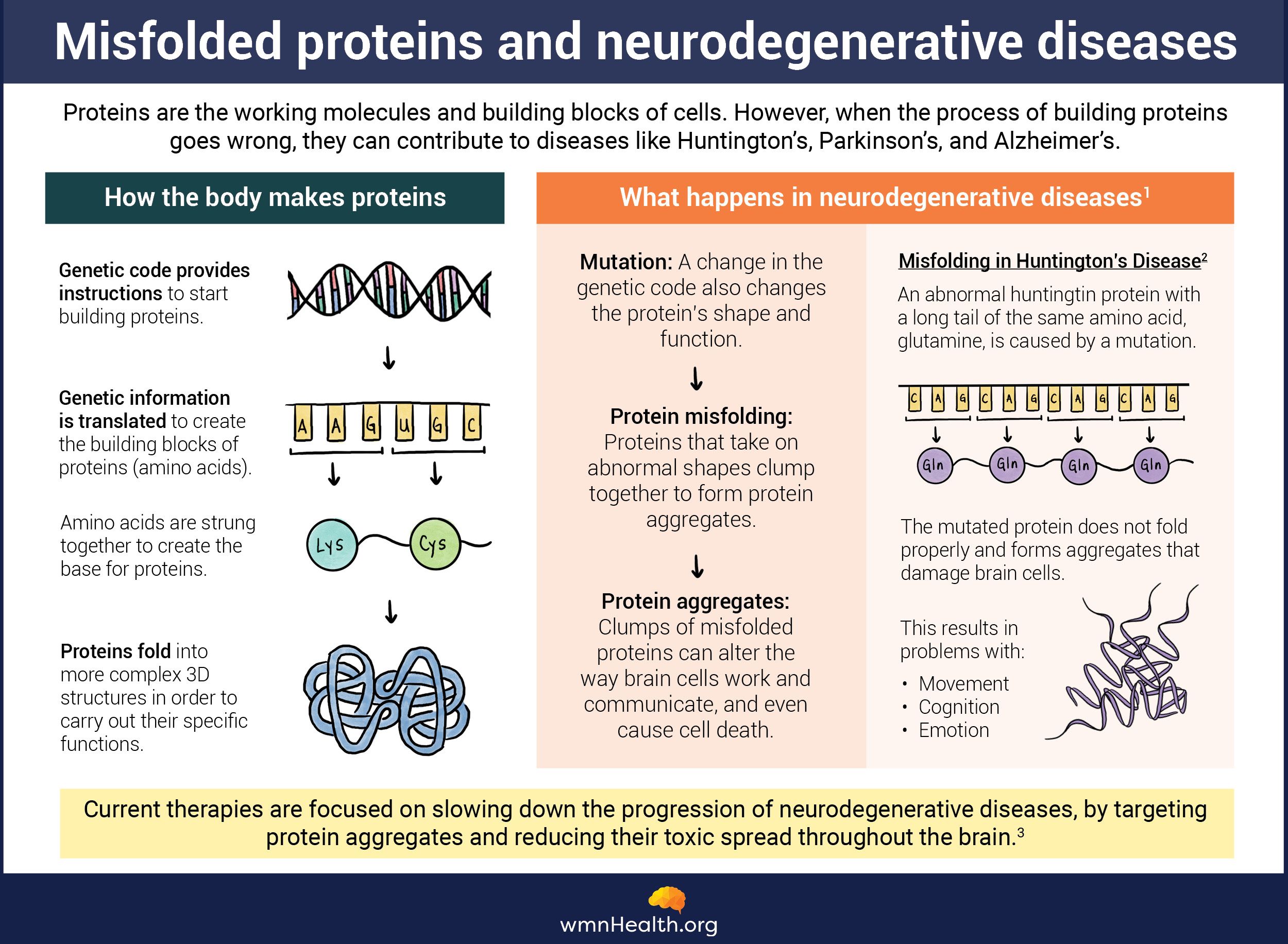
There are anywhere from 20,000 to 100,000 different proteins within a human cell, each one designated a specialized task. These complex molecules begin as long chains of amino acids that need to be arranged in a particular sequence to form functional structures.
Sometimes, however, proteins fail to fold into three-dimensional structures that work — instead turning into clumps or aggregates that cause cell damage. Protein-folding errors have been linked with a number of common neurodegenerative conditions, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and amyotrophic lateral sclerosis (ALS).
The misfolding proteins implicated in each of these conditions are different. However, experts believe understanding why these proteins fold into abnormal shapes and how they spread from cell to cell, often perpetuating disease, could help them develop strategies to slow down or halt the progression of these neurodegenerative diseases.
Why do proteins misfold, and how does that cause neurodegeneration?
Protein folding is a complex, haphazard process that is prone to error. A third of all newly synthesized proteins never make it to the final stage and are degraded and discarded by a cell’s quality control system. Sometimes, mutant proteins contort into unfavorable shapes and clump together like a messy ball of yarn, disrupting a cell’s ability to function. This accumulation of toxic protein clumps, experts believe, is at the heart of several common neurodegenerative disorders.
 Infographic by Cat Lau. (References below.)
Infographic by Cat Lau. (References below.)Huntington’s disease
Huntington’s disease is caused by the mutant huntingtin protein, which has an abnormally long string of a single amino acid called glutamine. A normal huntingtin protein contains anywhere from 10 to 34 glutamine repeats. However, when the repeats exceed 37, huntingtin molecules stick to one another and form tangled, rigid strands, says Jill Crittenden, molecular neurologist and scientific advisor at McGovern Institute for Brain Research at the Massachusetts Institute of Technology.
These strands, also known as fibrous protein aggregates, slowly accumulate within cells and disrupt their function. Studies have shown that huntington protein aggregates can also pass from one neuron to the next, spreading the disease and leading to its progression.
Huntington’s disease in females
Females with Huntington’s disease experience worse motor, cognitive, and depressive symptoms compared to men with the condition. These symptoms include:
- Muscle problems
- Slower eye movements
- Difficulty with speech
- Difficulty organizing tasks
- Slower thought processing
- Insomnia
- Social withdrawal
- Frequent thoughts of death
Source: “Sex Differences in Huntington's Disease: Evaluating the Enroll-HD Database” and Mayo Clinic.
Alzheimer’s and other dementias
Alzheimer’s disease is caused by a combination of proteins — amyloid beta and tau. Normally, tau is found inside a neuron’s axon, the hair-like cable that transmits signals from one neuron to the next. In Alzheimer’s and some other forms of dementia, tau proteins become damaged and clump together to form neurofibrillary tangles. These tangles disrupt communication between neurons and impair cognition, including memory and thought processing, which are the hallmarks of Alzheimer's.
On the other hand, the amyloid beta (or beta-amyloid) protein is normally found within the fatty membrane surrounding nerve cells and is important for neuronal growth and repair. But, when this naturally sticky protein is overproduced, it begins to form small clusters, called amyloid plaques, between brain cells. The plaques trigger an immune response that eventually leads to cell death.
 Infographic by Cat Lau. (References below.)
Infographic by Cat Lau. (References below.)Parkinson’s disease
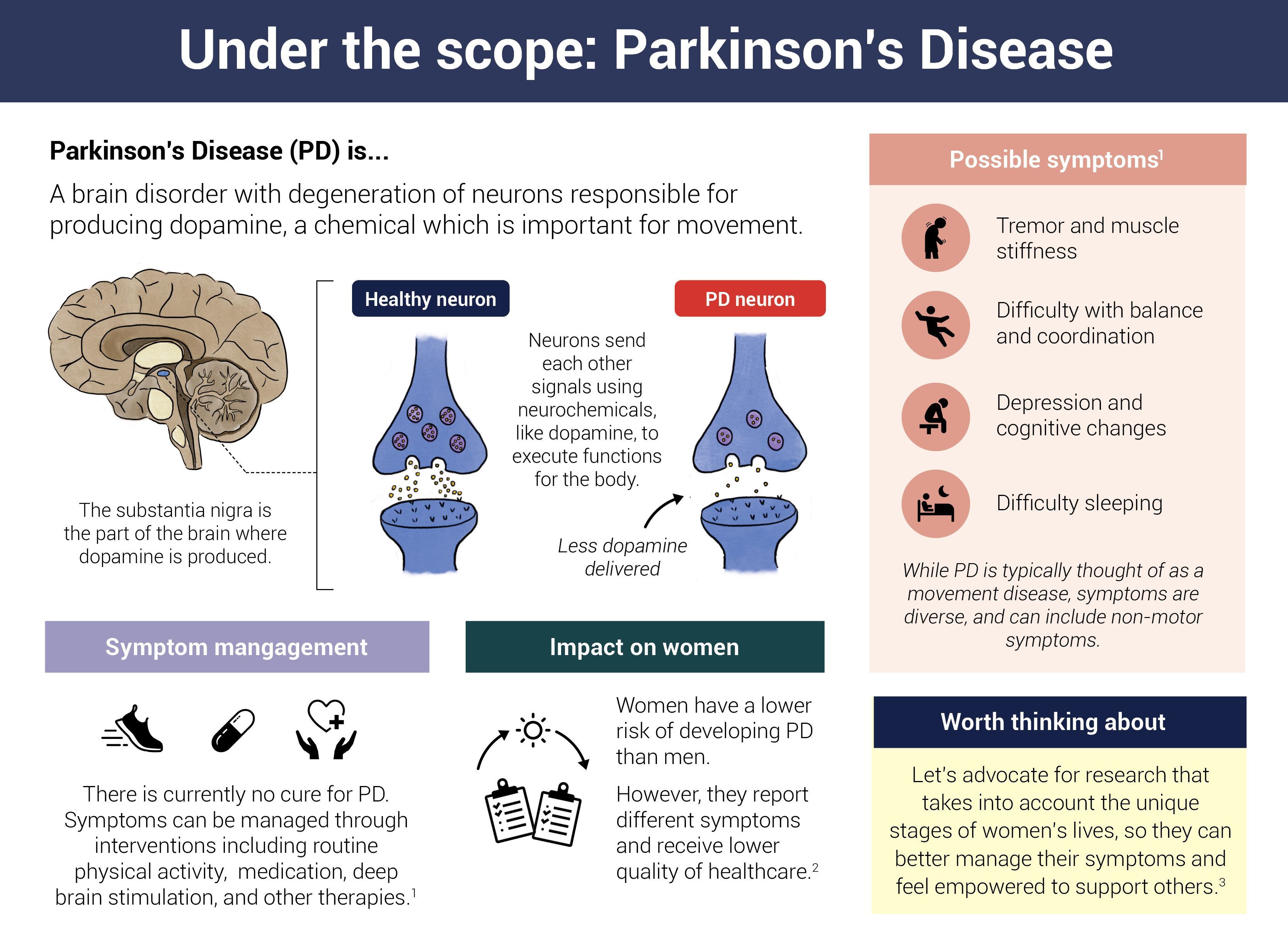
Parkinson’s disease is characterized by deposits of alpha-synuclein protein in the nervous system. When this protein that promotes crosstalk between brain cells misfolds, it forms clusters, also known as Lewy bodies, in the brain’s motor control areas/ The accumulation of Lewy bodies eventually kills off dopamine-producing neurons. The resulting dopamine deficiency gives rise to symptoms of Parkinson’s, like difficulty with movement and coordination, shaking, memory loss, and depression.
A recent study in mice suggests alpha-synuclein clumps can also accumulate in the gut and cause nearby proteins to misfold. These clumps then travel from the gut to the brain through the vagus nerve, a major signaling pathway between the brain and body.
 Infographic by Cat Lau. (References below.)
Infographic by Cat Lau. (References below.)Amyotrophic lateral sclerosis (ALS)
A number of misfolded proteins are linked to ALS, also known as Lou Gehrig’s disease, a progressive and debilitating disorder that leads to loss of muscle control and eventually death. These proteins include superoxide dismutase 1 (SOD1), TAR DNA-binding protein 43 (TDP-43), and FUsed in Sarcoma (FUS). These misfolded proteins can form aggregates in the brain’s motor cortex and spinal cord and spread to other regions over time.
Can rogue proteins be stopped?
Targeting misfolding proteins early enough in neurodegenerative diseases is critical for slowing their progression. Researchers have been honing in on the specific event, called nucleation, that starts off this chain reaction of protein misfolding, aggregation, and proliferation in the nervous system.
In a recent preprint, researchers at the Stowers Institute for Medical Research in Kansas City, Missouri, and their colleagues worked with yeast cells and found that if the abnormally long glutamine repeat (PolyQ) in huntingtin protein is stopped from aggregating early on, it can halt the neurodegenerative effects of Huntington’s disease. Interestingly, these findings have implications for similar conditions, like Alzheimer’s and Parkinson’s, that are set ablaze by misfolding and clumping of proteins.
Other experts are looking at the role of “chaperones” to stop rogue proteins from misfolding. This aptly named group of proteins assists others in folding properly and even offers misfolding proteins another chance to refold into a functional configuration. Studies have shown the benefit of chaperones in helping tau — the protein implicated in Alzheimer’s — get into its prescribed shape.
While there’s plenty that’s still unknown about neurodegenerative conditions and their progression, focusing on protein folding and misfolding mechanisms offers an interconnected path forward, where a piece in the puzzle of one neurodegenerative condition can help solve the mystery of the next.
Infographic references
Protein misfolding:
- Sweeney, P., Park, H., Baumann, M. et al. Protein misfolding in neurodegenerative diseases: implications and strategies. Transl Neurodegener 6, 6 (2017).
- What is Huntington's? Huntington's Disease News (Accessed November 10, 2023)
Alzheimer's
- 2023 Alzheimer's Facts and Figures: Special Report, Alzheimer's Association (Accessed June 20, 2023)
- Castro-Aldrete L, Moser MV, Putignano G, Ferretti MT, Schumacher Dimech A and Santuccione Chadha A (2023) Sex and gender considerations in Alzheimer’s disease: The Women’s Brain Project contribution. Front. Aging Neurosci. 15:1105620.
Parkinson's
- Parkinson's Disease, National Institutes of Health (Accessed June 9, 2023)
- Women and PD, Parkinsons.org (Accessed June 9, 2023)
- Subramanian, I., et al. Unmet Needs of Women Living with Parkinson's Disease: Gaps and Controversies. Movement Disorders, Vol. 37, No. 3, 2022